Enhanced methanol oxidation and oxygen reduction reactions on palladium-decorated FeCo@Fe/C core–shell nanocatalysts in alkaline medium
Abstract
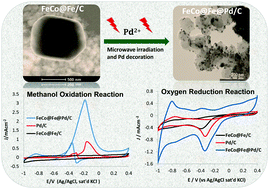
Palladium based nano-alloys are well known for their unique electrocatalytic properties. In this work, a palladium-decorated FeCo@Fe/C core–shell nanocatalyst has been prepared by a new method called microwave-induced top-down nanostructuring and decoration (MITNAD). This simple, yet efficient technique, resulted in the generation of sub-10 nm sized FeCo@Fe@Pd nanocatalysts (mainly 3–5 nm) from a micron-sized (0.21–1.5 μm) FeCo@Fe/C. The electrocatalytic activities of the core–shell nanocatalysts were explored for methanol oxidation reaction (MOR) and oxygen reduction reaction (ORR) in alkaline medium. A negative shift of 300 mV in the onset potential for MOR was observed, with a current thrice that of the Pd/C catalysts. A very low resistance to electron transfer (Rct) was observed while the ratio of forward-to-backward oxidation current (If/Ib) was doubled. The overpotential of ORR was significantly reduced with a positive shift of about 250 mV and twice the reduction current density was observed in comparison with Pd/C nanocatalysts with the same mass loading. The kinetic parameters (in terms of the Tafel slope (b) = −59.7 mV dec−1 (Temkin isotherm) and high exchange current density (jo) = 1.26 × 10−2 mA cm−2) provide insights into the favorable electrocatalytic performance of the catalysts in ORR in alkaline media. Importantly, the core–shell nanocatalyst exhibited excellent resistance to possible methanol cross-over during ORR, which shows excellent promise for application in direct alkaline alcohol fuel cells (DAAFCs).

 Please wait while we load your content...
Please wait while we load your content...