Factors affecting the selectivity of the photocatalytic conversion of nitroaromatic compounds over TiO2 to valuable nitrogen-containing organic compounds†
Abstract
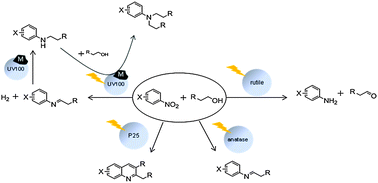
The photocatalytic conversion of various nitroaromatic compounds in alcohols employing four different types of TiO2 (Sachtleben Hombikat UV100 as anatase, Crystal Global R34 as rutile, Evonik-Degussa Aeroxide P25 as an anatase–rutile mixture, and home-made mesoporous anatase) has been studied. The effect of platinization of these different types of TiO2 on the reaction sequence has also been investigated. Upon irradiation of an ethanolic solution of m-nitrotoluene, as a model reaction, in the presence of the bare photocatalyst, different products were obtained according to the applied photocatalyst. It was found that the surface properties of the photocatalyst play an important role in the reaction pathway and thus in the selectivity of the products. In all cases, a simultaneous reduction of the nitro compound and an oxidation of the alcohol are induced by the photogenerated electrons and holes, respectively. An imine is then produced upon condensation of the generated aldehyde and amino compounds. Rutile was found to be more selective towards the primary amino compound (m-toluedine) while anatase catalysts gave a mixture of m-toluidine and its imine (N-ethylidene-3-methylaniline). A cyclization reaction of the produced imine to generate methyl quinoline was observed when Aeroxide P25 was used as a photocatalyst. Employing platinized TiO2, the hydrogenation of the produced imine yielding N-alkylated products was found to occur in all cases. However, the selectivity towards the mono N-alkylated product was the best using platinized Hombikat UV100. This selectivity was found to be also influenced by the loaded amount of Pt, the platinization method, and the illumination time but not by the light intensity.

 Please wait while we load your content...
Please wait while we load your content...