Microwave absorption in powders of small conducting particles for heating applications†
Abstract
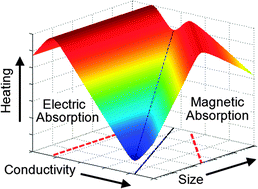
In microwave chemistry there is a common misconception that small, highly conducting particles heat profusely when placed in a large microwave electric field. However, this is not the case; with the simple physical explanation that the electric field (which drives the heating) within a highly conducting particle is highly screened. Instead, it is the magnetic absorption associated with induction that accounts for the large experimental heating rates observed for small metal particles. We present simple principles for the effective heating of particles in microwave fields from calculations of electric and magnetic dipole absorptions for a range of practical values of particle size and conductivity. For highly conducting particles, magnetic absorption dominates electric absorption over a wide range of particle radii, with an optimum absorption set by the ratio of mean particle radius a to the skin depth δ (specifically, by the condition a = 2.41δ). This means that for particles of any conductivity, optimized magnetic absorption (and hence microwave heating by magnetic induction) can be achieved by simple selection of the mean particle size. For weakly conducting samples, electric dipole absorption dominates, and is maximized when the conductivity is approximately σ ≈ 3ωε0 ≈ 0.4 S m−1, independent of particle radius. Therefore, although electric dipole heating can be as effective as magnetic dipole heating for a powder sample of the same volume, it is harder to obtain optimized conditions at a fixed frequency of microwave field. The absorption of sub-micron particles is ineffective in both magnetic and electric fields. However, if the particles are magnetic, with a lossy part to their complex permeability, then magnetic dipole losses are dramatically enhanced compared to their values for non-magnetic particles. An interesting application of this is the use of very small magnetic particles for the selective microwave heating of biological samples.

 Please wait while we load your content...
Please wait while we load your content...