Can self-assembly of copper(ii) picolinamide building blocks be controlled?†
Abstract
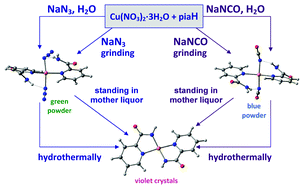
Four copper(II) derivatives with
* Corresponding authors
a
Laboratory of General and Inorganic Chemistry, Department of Chemistry, Faculty of Science, University of Zagreb, Horvatovac 102a, 10000 Zagreb, Croatia
E-mail:
mdjakovic@chem.pmf.hr, zpopovic@chem.pmf.hr
Fax: +385-1-4606-341
Tel: +385-1-4606-356
b
Departmento de Química e Bioquímica, CQB, Faculdade de Ciências, Universidade de Lisboa, Campo Grande, Ed. C8, 1749-049 Lisbon, Portugal
E-mail:
mjcalhorda@fc.ul.pt
Fax: +351-217500088
Tel: +351-217500196
c Fakulteta za kemijo in kemijsko tehnologijo, Univerza v Ljubljani, Aškerčeva c. 5, 1000 Ljubljana, Slovenia
d Inštitut za matematiko, fiziko in mehaniko, Jadranska c. 19, 1000 Ljubljana, Slovenia
e Fakulteta za gradbenuištvo in geodezijo, Univerza v Ljubljani, Jamova c. 2, 1000 Ljubljana, Slovenia
Four copper(II) derivatives with

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
M. Đaković, D. Vila-Viçosa, N. A. G. Bandeira, M. J. Calhorda, B. Kozlevčar, Z. Jagličić and Z. Popović, CrystEngComm, 2013, 15, 8074 DOI: 10.1039/C3CE41011K
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
