Hypothetical 3D-periodic covalent organic frameworks: exploring the possibilities by a first principles derived force field†
Abstract
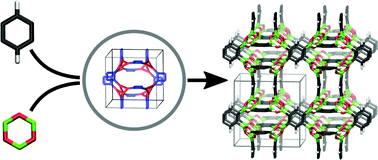
A scheme to predict as yet unknown, hypothetical covalent organic frameworks (COFs) from scratch by screening the possible space of supramolecular isomers is presented. This is achieved by extending our currently developed first principles derived force field MOF-FF with a parametrization for the boroxin unit. We considered four non-tetrahedral monomers with four boronic acid groups inspired by the corresponding carboxylate linkers known from metal–organic frameworks, and investigated the potential 3,4-connected topologies with edge-transitivity (ctn, bor, pto and tbo) or transitivity 32 (ofp, tfj, fjh, iab and nju). Due to the partly lower symmetry of the building blocks with respect to the vertex, beyond topological isomerism also isoreticular isomers are formed. We have used our reverse topological approach to construct the fictitious structures and employed an automated genetic algorithm based global minimum search approach to screen the vast configurational space of isoreticular isomerism and predicted a series of hypothetical 3D-COFs. All structures are completely relaxed by including the lattice parameters. From the atomistic structures, the accessible surface areas were determined, and, because of the isomer screening procedure, the question of crystallographic disorder could also be answered. Beyond the examples of hypothetical 3D-COFs serving as a lead for future synthetic investigations, this work is intended in particular to introduce the efficient predictive modeling method which can be applied to any kind of hypothetical COF system.
- This article is part of the themed collection: Covalent organic frameworks

 Please wait while we load your content...
Please wait while we load your content...