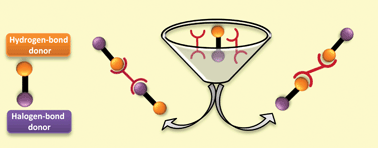
In order to study the structure-directing competition between hydrogen- and halogen-bond donors we have synthesized two ligands, 3,3′-azobipyridine and 4,4′-azobipyridine, and co-crystallized them with a series of bi-functional donor molecules comprising an activated halogen-bond donor (I or Br) as well as a hydrogen-bond donor (acid, phenol or oxime) on the same backbone. Based on the subsequent single-crystal analysis, 5 of 6 co-crystals of 3,3′-azobipyridine are assembled using hydrogen bonds as the primary driving force accompanied by weaker secondary (C–X⋯O) interactions. However, in 5 out of the 6 co-crystals of 4,4′-azobipyridine, both hydrogen bonds (O–H⋯N) and halogen bonds (C–X⋯N) are present as structure-directing interactions leading to 1-D chains. Since the charges on the acceptor sites in 3,3′- and 4,4′-azobipyridine are very similar, the observed difference in binding behaviour highlights the importance of binding-site location on the acceptor molecules (anti-parallel in 3,3′-azobipyridine and co-linear in 4,4′-azobipyridine) as a direct influence over the structural balance between hydrogen- and halogen-bond donors.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...