Nitrogenase: a general hydrogenator of small molecules
Abstract
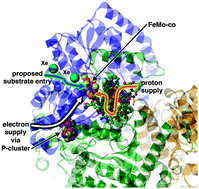
Nitrogenase naturally converts N2 to NH3, but it also hydrogenates a variety of small molecules, in many cases requiring multiple electrons plus protons for each catalytic cycle. A general mechanism, arising from many density functional calculations and simulations, is proposed to account for all of these reactions. Protons, supplied serially in conjunction with electrons to the active site FeMo-co (a CFe7MoS9 (homocitrate) cluster), generate H atoms that migrate over and populate two S and two Fe atoms in the reaction domain. The mechanistic paradigm is conceptually straightforward: substrate (on Fe) and H atoms (on S and Fe) are bound contiguously in the reaction zone, and H atoms transfer (probably with some quantum tunneling) to the substrate to form product. Details and justifications of the mechanisms for N2 and other key substrates are summarised, and the unusual structure of FeMo-co as a general hydrogenation catalyst is rationalised. Testing experiments are suggested.
- This article is part of the themed collection: Metal-Mediated Transformations of Small Molecules

 Please wait while we load your content...
Please wait while we load your content...