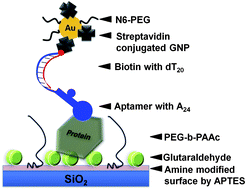
An evanescent-field-coupled waveguide-mode (EFC-WM) sensor utilizes monolithic SiO2/Si/SiO2 sensing plates having a multilayered structure and is used to evaluate a blocking agent comprising poly(ethylene glycol)-based block copolymers. Factor IX (FIX) protein was detected using its aptamer, viz. FIX was immobilized on a glutaraldehyde-modified silica surface, and then treated with a biotinylated aptamer. The quantitative analysis of FIX was carried out using streptavidin-conjugated gold nanoparticles (SA-GNPs). The blocking polymer, poly(ethylene glycol)-b-poly(acrylic acid) (PEG-b-PAAc), was found to mask unreacted amine and glutaraldehyde (Glu) moieties on the SiO2 surface, and it completely prevented the non-specific binding of SA-GNPs. By exploiting the strong blocking effect of PEG-b-PAAc, we achieved high ligand–analyte interaction sensitivity (sensitive down to 100 pM). To improve the sensitivity further, we also used pentaethylenehexamine-terminated PEG (N6-PEG) on GNPs. The improvement in sensitivity was found to be 1000-fold (to 100 fM), which was substantiated by the observation of higher numbers of GNPs on the sensing surface in the results of the scanning electron microscopic examination. Based on the competition assay of free biotin premixed with SA-GNPs, it was concluded that some active biotin-binding sites on the streptavidin were blocked by N6-PEG, which improved the binding ability to the biotinylated sensing surface. An optimum number of binding sites on the SA-GNPs might improve their binding affinity. The strategy shown with dual polymers, viz. blocking of the sensor chip surface and coating of SA-GNPs, is recommended for developing sensors with higher sensitivity and reliability. Selective binding of the aptamer to a very small amount of FIX in the mixed sample containing FXIa and FVIIa, or albumin, makes this the optimal strategy for detecting a FIX deficiency in human blood samples.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...