Can Janus particles give thermodynamically stable Pickering emulsions?
Abstract
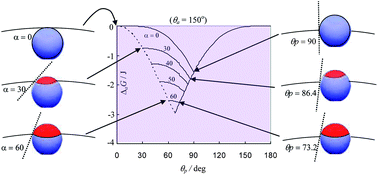
Emulsions stabilised by solid particles with homogeneous surfaces are thought to be kinetically rather than thermodynamically stable. In the absence of effects due to negative line tension in the contact line around adsorbed particles, the free energy of forming an emulsion droplet coated with a monolayer of adsorbed spherical particles is expected always to be positive, and is dominated by the presence of a fraction of uncovered oil–

 Please wait while we load your content...
Please wait while we load your content...