Cu2O|NiOx nanocomposite as an inexpensive photocathode in photoelectrochemical water splitting†
Abstract
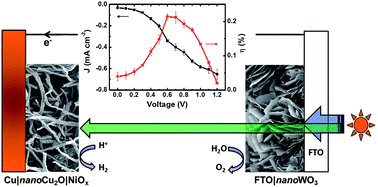
A photoelectrochemical (PEC) cell for overall water splitting made of a Cu2O nanowire photocathode modified with a thin film of NiOx coupled to a WO3 nanosheet photoanode is presented. The photocathode was prepared by thermal annealing of Cu(OH)2 nanowires on a Cu foil under N2, followed by the deposition of a 10 nm NiOx film on the Cu2O nanowires (aspect ratio > 40). XPS spectra revealed that the surface species of NiOx is a mixture of NiO and Ni(OH)2, which enhances charge separation in photoexcited Cu2O, as confirmed by electrochemical impedance spectroscopy. The optimized NiOx modified Cu2O electrode shows a photocurrent density up to −4.98 mA cm−2 at −0.33 V and −0.56 mA cm−2 at 0.1 V vs. the normal hydrogen electrode (NHE) under white-light irradiation (26 mW cm−2) in an aqueous electrolyte solution at pH 6 and 25 °C. The formation of H2 gas was only observed by gas chromatography for NiOx-modified Cu2O and was not detectable for unmodified Cu2O electrodes during prolonged irradiation. The nanocomposite structure also resulted in a three-fold increase in photostability of Cu2O; 72 ± 3% of the initial photocurrent density remained for the NiOx modified Cu2O electrode after 20 min irradiation at 0.1 V vs. NHE. The optimized photocathode was subsequently used in a two-electrode PEC cell with an n-type WO3 nanosheet photoanode for overall water splitting. The different band gap of Cu2O (2 eV) and WO3 (2.6 eV) permits for efficient and complementary light absorption and sunlight-driven water splitting. The p/n heterojunction PEC cell operates with a small output of electricity even in the absence of an external bias. We demonstrate that a Cu2O-based electrode for H2 evolution can be prepared free of noble metals and we show its utilization in a PEC water splitting cell made solely from earth abundant elements.

 Please wait while we load your content...
Please wait while we load your content...