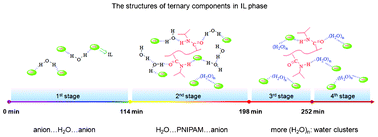
The structural evolution of payload transfer in biphasic systems is significant but difficult to accurately follow over the whole process. The dynamic transfer of homopolymer poly(N-isopropylacrylamide) (PNIPAM) from water to a hydrophobic IL (1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, [C2MIM][NTf2]) phase was investigated by in situ react infrared (IR) spectroscopy to reveal the detailed transfer process and structural changes of PNIPAM, water and [C2MIM][NTf2]. Compared with the corresponding binary systems, this work summarizes the four-step transfer behavior and the interactions among the components according to IR spectroscopy and multivariate curve resolution (MCR). At the initial stage, only some water diffuses into the hydrophobic IL in the form of a symmetric 1 : 2-type hydrogen bonded (anion⋯H2O⋯anion) species while most of the PNIPAM molecules are preserved in the water layer in the form of aggregates. Afterwards, the water molecules are bonded with PNIPAM in the aggregated state and they start to transfer together into the IL layer in the second stage where PNIPAM is changed into unfolding structures with complex interactions such as H2O⋯PNIPAM⋯anion, and meanwhile water structures of anion⋯(H2O)n⋯anion form due to further diffusion of water. After these stages, water molecules continue to dissolve in the IL although the polymer transfer is completed, and they are inclined to form further water clusters (H2O)n as deduced from the increase of OH region (3500–3300 cm−1) absorption. Finally, all of the components (PNIPAM, water and IL) coexist in the homogeneous IL phase with the aid of complex hydrogen bond and electric charge interactions.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...