An environmentally friendly method for the synthesis of nano-alumina with controllable morphologies
Abstract
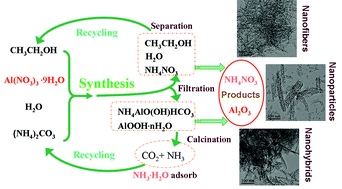
The relatively high price of aluminum alkoxides as an alumina source and organic surfactants as morphology control or structure directing agents are less attractive for practical applications to the synthesis of nanostructured alumina. Moreover, some of these synthesis processes may generate large masses of salt by-products. A highly efficient synthesis of nano-alumina from ammonium carbonate aqueous solution and aluminum nitrate ethanol solution without the use of a template and complicated processes has been achieved. The Al(NO3)3·9H2O reagent can be completely converted to nano-alumina without a by-product and the ethanol solutions can be reused. In addition, the CO2 and NH3 produced can also be recycled to the reagent (NH4)2CO3. The nano-alumina synthesized is of γ-alumina structure. Moreover, the simple and environmentally friendly route described could enhance the synthesis technology for nano-alumina and its applications.

 Please wait while we load your content...
Please wait while we load your content...