Regulatory function of divalent cations in controlling the acidogenic biohydrogen production process
Abstract
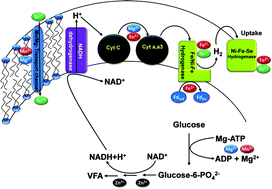
The functional role of divalent cations viz., iron (Fe2+), magnesium (Mg2+), nickel (Ni2+), zinc (Zn2+) and manganese (Mn2+) in regulating the biohydrogen (H2) production potential of a biocatalyst was evaluated under varying concentrations. The optimum conditions were evaluated in detail and compared with the principle conditions for enhanced conversion efficiency. Higher concentration of Fe2+ showed an enhanced H2 production efficiency of the biocatalyst (61.94 mmol) due to its role as a component of hydrogenase and ferredoxin. On the contrary, a higher concentration of Mg+2 showed a higher substrate utilization capability (76.46%) of the biocatalyst. The optimum metal concentration showed a higher H2 yield (18.23 mol kg−1 CODR; 3.5 mol mol−1 glucose) over principle operation conditions (14.76 mol kg−1 CODR; 2.84 mol mol−1 glucose). Higher concentrations of acetate (78 ± 6%) as the metabolic intermediate over butyrate (12 ± 2%) supported the observed higher H2 yields. Higher reductive catalytic current over oxidative current along with the enhanced dehydrogenase activity supported the higher H2 production efficiency. Shift in the redox Tafel slope was witnessed near zero in the case of Fe2+ and optimum conditions indicating the simultaneous oxidation and reduction reactions facilitating the availability of higher number of protons through metabolite inter-conversions that can make H2. The presence of divalent cations at an optimum concentration will enhance the H2 production capability of the biocatalyst. However, elevated levels of metal concentration showed a negative influence on the biocatalyst efficiency.

 Please wait while we load your content...
Please wait while we load your content...