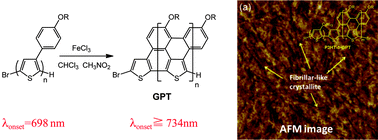
Novel polythiophenes with graphene structures (GPTs) could be synthesized based on intramolecular oxidative coupling of the polythiophene precursors having phenyl groups at the 3-position, poly(3-(4′-(3′′,7′′-dimethyloctoxy)phenyl)thiophene) (P3PhT) and poly(3-hexylthiophene)-b-P3PhT (P3HT-b-P3PhT), using FeCl3 as an oxidative agent. The GPTs were characterized by 1H NMR spectroscopy, UV-vis spectroscopy, FT-IR spectroscopy, cyclic voltammetry and X-ray diffraction. The thin film transistor (TFT) characteristics were also performed for P3HT-b-GPT. The progression of oxidative coupling of P3PhTs was confirmed by 1H NMR and FT-IR spectroscopies. Moreover, GPTs showed significant red-shifts in the UV-vis spectra due to their high coplanarity, extended π-conjugation length, and strong π-stacking formation. P3HT-b-GPT also showed a red-shift in the UV-vis spectra and a reduced band gap as compared to the pristine P3HT. P3HT-b-GPT exhibited hole mobility (∼10−5 cm2 V−1 s−1) which is comparable to that of pristine P3HT having a similar molecular weight.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...