Synthesis, photophysical, photochemical, DNA cleavage/binding and cytotoxic properties of pyrene oxime ester conjugates†
Abstract
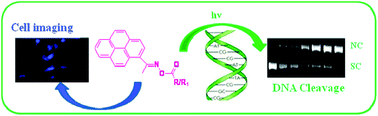
A new series of (E)-pyrene oxime ester conjugates of carboxylic acids including amino acids were synthesized by coupling with an environment sensitive fluorophore 1-acetylpyrene. (E)-Pyrene oxime esters exhibited strong fluorescence properties and interestingly their fluorescence properties were found to be highly sensitive to the surrounding environment. Direct irradiation of the (E)-pyrene oxime esters by UV light (≥350 nm) resulted in both the photo-Beckmann rearrangement product and products resulting from N–O bond homolysis. Photoproduct formation and their distribution were found to be solvent dependent. Further, we also showed (E)-pyrene oxime esters intercalated into DNA efficiently and photo-cleaved DNA. Finally we also showed these oxime esters can permeate cells efficiently and may cause cytotoxicity upon irradiation of light.

 Please wait while we load your content...
Please wait while we load your content...