Photochemical oxazole–nitrile conversion downstream of rhizoxin biosynthesis and its impact on antimitotic activity†‡
Abstract
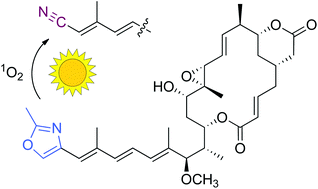
Through metabolic profiling of mutants and wild type of the endofungal bacterium Burkholderia rhizoxinica two novel rhizoxin derivatives with unusual
- This article is part of the themed collection: Organic & Biomolecular Chemistry 10th Anniversary

 Please wait while we load your content...
Please wait while we load your content...