First diastereoselective synthesis of methyl caffeoyl- and feruloyl-muco-quinates†
Abstract
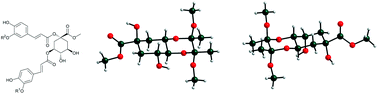
We report on a diastereoselective synthesis of six derivatives of caffeoyl- and feruloyl-muco-quinic acids. All the muco-quinic acid derivatives were obtained in excellent yield in five steps starting from

 Please wait while we load your content...
Please wait while we load your content...