Unique photocatalytic oxidation reactivity and selectivity of TiO2–graphene nanocomposites
Abstract
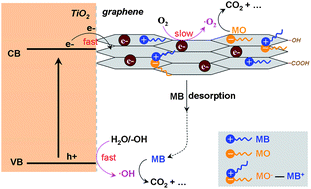
Mesoporous TiO2–graphene nanocomposites are fabricated in high yield via two successive steps: (1) hydrothermal hydrolysis of Ti(SO4)2 in an acidic suspension of graphene oxide to gain TiO2–graphene oxide nanocomposites; (2) UV-assisted photocatalytic reduction of graphene oxide to get the TiO2–graphene nanocomposites. The anatase TiO2 nanocrystals with a crystallite size of 10–20 nm are densely packed and supported on meshy graphene sheets with close interfacial contacts, which is confirmed by transmission electron microscopy (TEM) together with Raman spectroscopy and X-ray photoelectron spectroscopy (XPS). Although a low graphene loading (0–2 wt%) slightly influences the textural properties (including the crystallite size, specific surface areas, and pore volume etc.), the incorporation of graphene in TiO2–graphene nanocomposites greatly increases the adsorption capacity towards azo dyes such as MO and MB, which is possibly associated with their unique surface properties. Significantly, the incorporated graphene exerts combined effects on the adsorption and charge transfer dynamics in TiO2–graphene nanocomposites, which together endow them with good photocatalytic reactivity and tunable photocatalytic selectivity in decomposing MO and MB in aqueous solution.

 Please wait while we load your content...
Please wait while we load your content...