A matched molecular pair analysis of in vitro human microsomal metabolic stability measurements for heterocyclic replacements of di-substituted benzene containing compounds – identification of those isosteres more likely to have beneficial effects†
Abstract
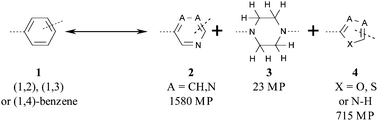
A matched molecular pair analysis (MMPA) was used to study the classic medicinal chemistry transformation of a di-substituted

 Please wait while we load your content...
Please wait while we load your content...