Bright, emission tunable fluorescent dyes based on imidazole and π-expanded imidazole†
Abstract
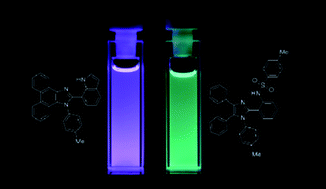
A diverse set of imidazole- and π-expanded imidazole derivatives displaying excited state intramolecular proton transfer (ESIPT) was designed and synthesized. The effect of structural variation on photophysical properties was studied in detail for nine dyes. The relationship between the structure and photophysical properties was thoroughly elucidated also by comparing with analogues with blocked ESIPT functionality. All but one of the obtained compounds exhibit ESIPT, as demonstrated by large Stokes shifts (6500–15 600 cm−1). The type of π-expansion strongly influences the overall optical phenomena: while typical π-expansion preserves ESIPT activity, the direct fusion of imidazole with a naphthalene unit at positions 4 and 5 results in dyes which do not exhibit ESIPT. The compound possessing an acidic NH group as part of an intramolecular hydrogen bond system has a much higher fluorescence quantum yield and Stokes shift than its analogue bearing an OH group. The occurrence of ESIPT for tosylamide analogues is less affected by the hydrogen-bonding ability of the solvents compared to the unprotected amines. Two-photon absorption cross-sections of the selected derivatives are in the range of 5–100 GM.

 Please wait while we load your content...
Please wait while we load your content...