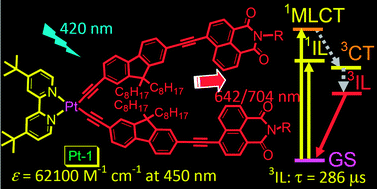
Fluorene-containing N^N Pt(II) bisacetylide complexes were prepared, in which the fluorene moieties were connected to the Pt(II) center via acetylide bonds (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–). Different aryl groups were attached to the fluorene moiety, such as phenylacetylide (Pt-4), naphthalimide-4-acetylide (Pt-1) and in Pt-2, the fluorene moiety was changed to carbozale moiety. We found that with the fluorene linker between the arylacetylide and the Pt(II) center, the absorption of complexes in the visible range were intensified. All the complexes show room temperature (RT) phosphorescence. Furthermore, Pt-1 shows much longer triplet excited state lifetime (τ = 138.1 μs) than the analogue Pt-3 (τ = 47.4 μs). For Pt-2, with changing the fluorene moiety to carbazole moiety, the T1 state lifetime becomes much shorter (τ = 23.0 μs). Thus the one-atom (N vs. C) difference is crucial for the photophysical properties. The triplet excited state of Pt-1 was proved to be the intraligand excited state (3IL) by nanosecond time-resolved transient absorption spectroscopy, spin density analysis and emission at 77 K. The complexes were used as triplet sensitizers for triplet–triplet annihilation (TTA) upconversion. Upconversion quantum yield (ΦUC) up to 24.3% was observed for Pt-1. Under the same conditions the model complex Pt-4 shows no upconversion. The overall upconversion efficiency (η) of the new complexes are improved compared to the model complexes, such as Pt-2, Pt-3 and Pt-4. The improved upconversion efficiency was attributed to either the prolonged T1 excited state lifetime or the intensified absorption in the visible range. Our study on the fluorene-containing N^N Pt(II) bisacetylide complexes will be useful for designing new phosphorescent Pt(II) complexes and for their applications.
C–). Different aryl groups were attached to the fluorene moiety, such as phenylacetylide (Pt-4), naphthalimide-4-acetylide (Pt-1) and in Pt-2, the fluorene moiety was changed to carbozale moiety. We found that with the fluorene linker between the arylacetylide and the Pt(II) center, the absorption of complexes in the visible range were intensified. All the complexes show room temperature (RT) phosphorescence. Furthermore, Pt-1 shows much longer triplet excited state lifetime (τ = 138.1 μs) than the analogue Pt-3 (τ = 47.4 μs). For Pt-2, with changing the fluorene moiety to carbazole moiety, the T1 state lifetime becomes much shorter (τ = 23.0 μs). Thus the one-atom (N vs. C) difference is crucial for the photophysical properties. The triplet excited state of Pt-1 was proved to be the intraligand excited state (3IL) by nanosecond time-resolved transient absorption spectroscopy, spin density analysis and emission at 77 K. The complexes were used as triplet sensitizers for triplet–triplet annihilation (TTA) upconversion. Upconversion quantum yield (ΦUC) up to 24.3% was observed for Pt-1. Under the same conditions the model complex Pt-4 shows no upconversion. The overall upconversion efficiency (η) of the new complexes are improved compared to the model complexes, such as Pt-2, Pt-3 and Pt-4. The improved upconversion efficiency was attributed to either the prolonged T1 excited state lifetime or the intensified absorption in the visible range. Our study on the fluorene-containing N^N Pt(II) bisacetylide complexes will be useful for designing new phosphorescent Pt(II) complexes and for their applications.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–). Different aryl groups were attached to the fluorene moiety, such as
C–). Different aryl groups were attached to the fluorene moiety, such as 

 Please wait while we load your content...
Please wait while we load your content...