Formation of reactive aerogels and their reactivity in aqueous media. Wettability induces hydrophobic vs. hydrophilic selectivity†
Abstract
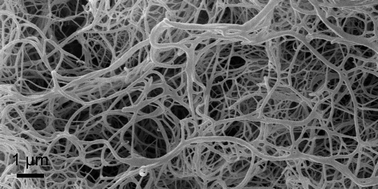
Aerogels were formed from organogels of diamides by a supercritical drying process. The used organogels are composed of self-assembled

 Please wait while we load your content...
Please wait while we load your content...