Mechanical compaction directly modulates the dynamics of bile canaliculi formation†
Abstract
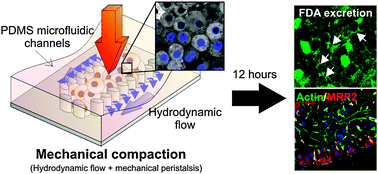
Homeostatic pressure-driven compaction is a ubiquitous mechanical force in multicellular organisms and is proposed to be important in the maintenance of multicellular tissue integrity and function. Previous cell-free biochemical models have demonstrated that there are cross-talks between compaction forces and tissue structural functions, such as cell–cell adhesion. However, its involvement in physiological tissue function has yet to be directly demonstrated. Here, we use the bile canaliculus (BC) as a physiological example of a multicellular functional structure in the liver, and employ a novel 3D microfluidic hepatocyte culture system to provide an unprecedented opportunity to experimentally modulate the compaction states of primary hepatocyte aggregates in a 3D physiological-mimicking environment. Mechanical compaction alters the physical attributes of the hepatocyte aggregates, including cell shape, cell packing density and cell–cell contact area, but does not impair the hepatocytes' remodeling and functional capabilities. Characterization of structural and functional polarity shows that BC formation in compact hepatocyte aggregates is accelerated to as early as 12 hours post-seeding; whereas non-compact control requires 48 hours for functional BC formation. Further dynamic immunofluorescence imaging and gene expression profiling reveal that compaction accelerated BC formation is accompanied by changes in actin cytoskeleton remodeling dynamics and transcriptional levels of hepatic nuclear factor 4α and Annexin A2. Our report not only provides a novel strategy of modeling BC formation for in vitro hepatology research, but also shows a first instance that homeostatic pressure-driven compaction force is directly coupled to the higher-order multicellular functions.

 Please wait while we load your content...
Please wait while we load your content...