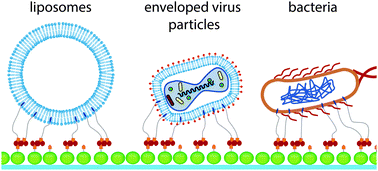
Immobilisation of liposomes and cells is often a prerequisite for long-term observations. The most common immobilisation approaches rely on surface modifications, encapsulation in porous materials or trapping in microfluidic channels by means of hurdle-like structures. While these approaches are useful for larger mammalian cells, the immobilisation of smaller organisms like bacteria and yeast or membrane model systems such as liposomes typically requires modification of their outer membrane to ensure that they are stably arrested at a defined position. Here, we present a protocol to immobilise biological objects, which can interact with hydrophobic cholesterol. A water-soluble molecule (cholesterol–PEG–biotin) is used as a linker, which can bind via avidin to biotinylated BSA (bBSA) previously absorbed on a glass surface. For better visualization, bBSA is arranged in a dot pattern by means of microcontact printing, and a microfluidic channel is used for sample supply. We show that our approach can be used to successfully immobilise artificial liposomes of different sizes, native (cell-derived) vesicles, vaccinia virions, Saccharomyces cerevisiae and Escherichia coli, simply by flushing the objects through the channel. Under these conditions, small liposomes and biological objects are stably arrested at high flow rates, while larger cells and liposomes can be released again by application of high shear stress. This protocol can be applied for long-term studies where fluids must be changed repeatedly, for measuring fast kinetics where rapid fluid exchange is essential, and to study the effects of shear stress.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...