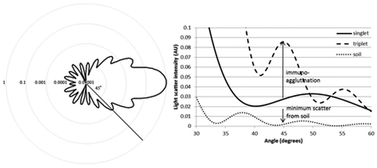
Cryptosporidium spp. is an obligate, parasitic protozoan that is difficult to detect and causes diarrhea in healthy adults while potentially causing death in the immunocompromised and children. Its treatment options are few and treat the symptoms, not the actual parasite. Current methods of detection are inefficient and rely too heavily upon laboratory sample preparations and technician skill, including differential staining, negative staining, and immunofluorescence methods [especially U.S. Environmental Protection Agency (EPA) Method 1623]. These assays can take from hours to days and require a laboratory environment. In this work, we demonstrated the microbead immunoagglutination assay combined with Mie scatter detection in a microfluidic device to provide a field-deployable and near-real-time alternative to the laboratory-based method (especially EPA Method 1623). Two main challenges were the relatively big diameter of Cryptosporidium oocysts (5–6 μm) and the contaminants in field water samples that negatively affected the immunoagglutination and its scatter detection. We used 4 min sonication to liberate Cryptosporidium oocyst wall proteins (COWP), which was previously used to inactivate Cryptosporidium oocysts. As for the contaminants, we optimized the microbead diameter (920 nm) and the wavelength of incident light (375 nm) to find the angle of scatter detection (45°) where the Mie scatter from immunoagglutinated microbeads was maximum and the background scatter from contaminants was minimum. This enabled the sub-single-oocyst-level detection despite the fact that only a very small volume of water sample (15 μL) was introduced to the microfluidic biosensor. When combined with filtration/concentration, this method is able to detect ≤1 oocyst per large volume of water, comparable to or potentially better than the EPA method 1623, while effectively reducing the time and labor necessary for staining and microscopic analysis. For faster, near-real-time assays, filtration/concentration may not be used, where the detection limit was 1–10 oocysts per mL with the total assay time of 10 min including the 4 min sonication time. The linear range of assay was over 5 orders of magnitude. The final device was compact and had the potential to be used in field situations, and required less technical expertise and/or training compared to the other methods.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...