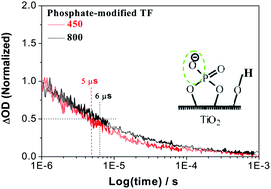
Phosphate modified nanocrystalline TiO2 (nc-TiO2) films were prepared by a doctor blade method, followed by post-treatment with monometallic sodium orthophosphate solution. The dynamic processes of the photogenerated charges from the resulting nc-TiO2 films were thoroughly investigated by means of transient absorption spectroscopy (TAS). It is shown that photogenerated holes in the un-modified TiO2 film exhibit the same dynamic decay process as its photogenerated electrons, in oxygen-free water of pH 7. However, photogenerated holes in the phosphate modified film display a slightly faster dynamic decay process than its photogenerated electrons, and photogenerated charges of the modified film have a much longer lifetime than those of the un-modified film. These differences are attributed to the surface-carried negative charges of nc-TiO2 resulting from the phosphate groups (–Ti–O–P–O−). Interestingly, the photoelectrochemical (PEC) experiments show that modification with an appropriate amount of phosphate could improve the photocurrent density of the nc-TiO2 film electrode by about 2 times, at a voltage of 0 V in the neutral electrolyte. Based on the TAS and PEC measurements of un-modified and phosphate modified nc-TiO2 films, with different conditions, it is suggested that the prolonged lifetime of photogenerated charges can be attributed to the negative electrostatic field formed in the surface layers. It is also responsible for the increase in activity for PEC water splitting and for the reported photocatalytic degradation of pollutants. The suggested mechanism would be applicable to other oxide semiconductor photocatalysts and to modification with other inorganic anions.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...