The role of proton coupled electron transfer in water oxidation†
Abstract
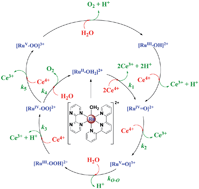
Water oxidation is a key half reaction in energy conversion schemes based on solar fuels and targets such as light driven water splitting or carbon dioxide reduction into CO, other oxygenates, or hydrocarbons. Carrying out these reactions at rates that exceed the rate of solar insolation for the extended periods of time required for useful applications presents a major challenge. Water oxidation is the key “other” half reaction in these schemes and it is dominated by PCET given its multi-electron, multi-proton character, 2H2O → O2 + 4e− + 4H+. Identification of PCET was an offshoot of experiments designed to investigate energy conversion by electron transfer quenching of molecular excited states. The concepts “redox potential leveling” and concerted electron–proton transfer came from measurements on stepwise oxidation of cis-RuII(bpy)2(py)(OH2)2+ to RuIV(bpy)2(py)(O)2+. The Ru “blue dimer”, cis,cis-(bpy)2(H2O)RuORu(OH2)(bpy)24+, was the first designed catalyst for water oxidation. It undergoes oxidative activation by PCET to give the transient (bpy)2(O)RuVORuV(O)(bpy)24+, O-atom attack on water to give a peroxidic intermediate, and further oxidation and O2 release. More recently, a class of single site water oxidation catalysts has been identified, e.g., Ru(tpy)(bpm)(OH2)2+ (tpy is 2,2′:6′,2′′-terpyridine; bpm is 2,2′-bipyrimidine). They undergo stepwise PCET oxidation to RuIV=O2+ or RuV(O)3+ followed by O-atom transfer with formation of peroxidic intermediates which undergo further oxidation and O2 release. PCET plays a key role in the three zones of water oxidation reactivity: oxidative activation, O⋯O bond formation, oxidation and O2 release from peroxidic intermediates. Similar schemes have been identified for electrocatalytic water oxidation on oxide electrode surfaces based on phosphonated derivatives such as [Ru(Mebimpy)(4,4′-(PO3H2CH2)2bpy)(OH2)]2+. A PCET barrier to RuIII–OH2+ → RuIV=O2+ oxidation arises from the large difference in pKa values between RuIII–OH2+ and RuIV(OH)3+. On oxide surfaces this oxidation occurs by multiple pathways. Kinetic, mechanistic, and DFT results on single site catalysts reveal a new pathway for the O⋯O bond forming step (Atom-Proton Transfer, APT), significant rate enhancements by added proton acceptor bases, and accelerated water oxidation in propylene carbonate as solvent with water added as a stoichiometric reagent. Lessons learned about water oxidation and the role of PCET and concerted pathways appear to have direct relevance for water oxidation in Photosystem II (PSII) with PSII a spectacular example of PCET in action. This includes a key role for Multiple Site-Electron Proton Transfer in oxidative activation of the Oxygen Evolving Complex (OEC) in the S0 → S1 transition in the Kok cycle.

 Please wait while we load your content...
Please wait while we load your content...