Using combinations of oxidants and bases as PCET reactants: thermochemical and practical considerations†
Abstract
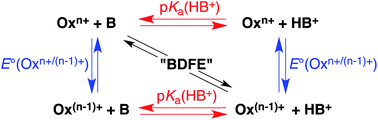
Studies in proton-coupled electron transfer (PCET) often require the combination of an outer-sphere oxidant and a base, to remove an electron and a proton. A common problem is the incompatibility of the oxidant and the base, because the former is electron deficient and the latter electron rich. We have tested a variety of reagents and report a number of oxidant/base combinations that are compatible and therefore potentially useful as PCET reagents. A formal bond dissociation free energy (BDFE) for a reagent combination is defined by the redox potential of the oxidant and pKa of the base. This is a formal BDFE because no X–H bond is homolytically cleaved, but it is a very useful way to categorize the H• accepting ability of an oxidant/base PCET pair. Formal BDFEs of stable oxidant/base combinations range from 71 to at least 98 kcal mol−1. Effects of solvent, concentration, temperature, and counterions on the stability of the oxidant/base combinations are discussed. Extensions to catalysis and related reductant/acid combinations are mentioned.

 Please wait while we load your content...
Please wait while we load your content...