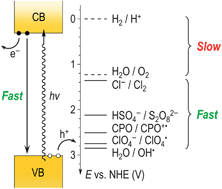
The faradaic efficiency for O2(g) evolution at thin-film WO3 photoanodes has been evaluated in a series of acidic aqueous electrolytes. In 1.0 M H2SO4, persulfate was the predominant photoelectrochemical oxidation product, and no O2 was detected unless catalytic quantities of Ag+(aq) were added to the electrolyte. In contact with 1.0 M HClO4, dissolved O2 was observed with nearly unity faradaic efficiency, but addition of a hole scavenger, 4-cyanopyridine N-oxide, completely suppressed O2 formation. In 1.0 M HCl, Cl2(g) was the primary oxidation product. These results indicate that at WO3 photoanodes, water oxidation is dominated by oxidation of the acid anions in 1.0 M HCl, H2SO4, and HClO4, respectively.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...