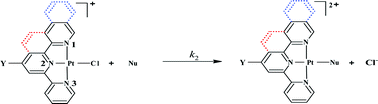
The nucleophilic substitution reactions of complexes [Pt{4′-(2′′′-CH3-phenyl)-2,2′:6′,2′′-terpyridine}Cl]CF3SO3, [CH33PhPtCl], [Pt{4′-(2′′′-CH3-phenyl)-6-(3′′-isoquinoyl)-2,2′bipyridine}Cl]SbF6, [CH33PhisoqPtCl], [Pt{2-(2′-pyridyl)-1,10-phenanthroline}Cl]Cl, [pyPhenPtCl], and [Pt(terpyridine)Cl]+, [PtCl] with a series of nucleophiles: thiourea (TU), N,N-dimethylthiourea (DMTU), N,N,N,N-tetramethylthiourea (TMTU), I−, Br−, and SCN− were studied in 0.1 M LiCF3SO3 in methanol (in the presence of 10 mM LiCl). The reactivity of the investigated complexes follows the order pyPhenPtCl > PtCl > CH33PhPtCl > CH33PhisoqPtCl. The lability of the chloride ligand is dependent on the strength of π-backbonding properties of the spectator ligands around the platinum centre. The experimental data is strongly supported by DFT calculations. The dependence of the second-order rate constants on concentration of the nucleophiles as well as the large negative values reported for the activation entropy (ΔS‡) confirmed an associative mechanism of substitution.

 Please wait while we load your content...
Please wait while we load your content...