Effect of chelating ring size in catalytic ketone hydrogenation: facile synthesis of ruthenium(ii) precatalysts containing an N-heterocyclic carbene with a primary amine donor for ketone hydrogenation and a DFT study of mechanisms†
Abstract
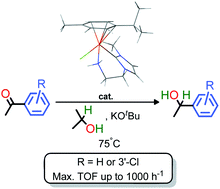
A half-sandwich ruthenium(II) complex, [Ru(η6-p-cymene)(C–NH2)Cl]PF6 (4·PF6), containing an N-heterocyclic carbene (NHC) with a primary amine donor (C–NH2) which chelates through the carbene carbon and the amine nitrogen to form a 6-membered ring was synthesized in a one-pot reaction starting from a primary-amine functionalized imidazolium salt 2. Complex 4·PF6 catalyzed the hydrogenation of ketones using 2-propanol or H2 as the reductant. A maximum turnover frequency of 1062 h−1 and a turnover number of 1140 at 5 h were achieved for the transfer hydrogenation of 3′-chloroacetophenone in 2-propanol at 75 °C. A cationic hydride–amine complex 5, [Ru(η6-p-cymene)(C–NH2)H]PF6, was synthesized, and this reacted very slowly with acetophenone unless first activated by an alkoxide base. Computational studies by DFT methods suggested that the poor reactivity of the hydride–amine complex 5 was attributed to a large barrier for the transfer of its H+/H− couple to a ketone for bifunctional catalysis. An inner-sphere mechanism, which involves a decoordinated amine group of the C–NH2 ligand, was computed to be a feasible energetic pathway in comparison to the computed outer-sphere bifunctional mechanism. This explains the catalytic activity and selectivity that is observed for the newly synthesized ruthenium(II) catalysts.

 Please wait while we load your content...
Please wait while we load your content...