The consecutive syntheses of imidazoles 1-(4-X-C6H4)-4,5-R2-cC3HN2 (3a, X = Br, R = H; 3b, X = I, R = Me; 3c, X = H, R = Me; 5, X = Fc, R = H; 7, X = C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CFc, R = H; 9, X = C6H5, R = Me; Fc = Fe(η5-C5H4)(η5-C5H5)), phosphino imidazoles 1-(4-X-C6H4)-2-PR′2-4,5-R2-cC3N2 (11a–k; X = Br, I, Fc, FcC
CFc, R = H; 9, X = C6H5, R = Me; Fc = Fe(η5-C5H4)(η5-C5H5)), phosphino imidazoles 1-(4-X-C6H4)-2-PR′2-4,5-R2-cC3N2 (11a–k; X = Br, I, Fc, FcC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C, Ph; R = H, Me; R′ = Ph, cC6H11, cC4H3O), imidazolium salts [1-(4-X-C6H4)-3-R′′-4,5-R2-cC3HN2]I (16a; X = Br, R = H, R′′ = n-Bu; 16b, X = Br, R = H, R′′ = n-C8H17; 16c, X = I, R = Me, R′′ = n-C8H17, 16d, X = H, R = Me, R′′ = n-C8H17) and phosphino imidazolium salts [1-C6H5-2-PR′2-3-n-C8H17-4,5-Me2-cC3N2]PF6 (17a, R′ = C6H5; 17b, R′ = cC6H11) or [1-(4-P(C6H5)2-C6H4)-3-n-C8H17-4,5-Me2-cC3HN2]PF6, (20) and their selenium derivatives 1-(4-X-C6H4)-2-P(
C, Ph; R = H, Me; R′ = Ph, cC6H11, cC4H3O), imidazolium salts [1-(4-X-C6H4)-3-R′′-4,5-R2-cC3HN2]I (16a; X = Br, R = H, R′′ = n-Bu; 16b, X = Br, R = H, R′′ = n-C8H17; 16c, X = I, R = Me, R′′ = n-C8H17, 16d, X = H, R = Me, R′′ = n-C8H17) and phosphino imidazolium salts [1-C6H5-2-PR′2-3-n-C8H17-4,5-Me2-cC3N2]PF6 (17a, R′ = C6H5; 17b, R′ = cC6H11) or [1-(4-P(C6H5)2-C6H4)-3-n-C8H17-4,5-Me2-cC3HN2]PF6, (20) and their selenium derivatives 1-(4-X-C6H4)-2-P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se)R′2-4,5-R2-cC3N2 (11a-Se–f-Se; X = Br, I; R = H, Me; R′ = C6H5, cC6H11, cC4H3O) are reported. The structures of 11a-Se and [(1-(4-Br-C6H4)-cC3H2N2-3-n-Bu)2PdI2] (19) in the solid state were determined. Cyclovoltammetric measurements were performed with the ferrocenyl-containing molecules 5 and 7 showing reversible redox events at E0 = 0.108 V (ΔEp = 0.114 V) (5) and E0 = 0.183 V (ΔEp = 0.102 V) (7) indicating that 7 is more difficult to oxidise. Imidazole oxidation does not occur up to 1.3 V in dichloromethane using [(n-Bu)4N][B(C6F5)4] as supporting electrolyte, whereas an irreversible reduction is observed between −1.2–−1.5 V. The phosphino imidazoles 11a–k and the imidazolium salts 17a,b and 20, respectively, were applied in the Suzuki C–C cross-coupling of 2-bromo toluene with phenylboronic acid applying [Pd(OAc)2] as palladium source. Depending on the electronic character of 11a–k, 17a,b and 20 the catalytic performance of the in situ generated catalytic active species can be predicted. As assumed, more electron-rich phosphines with their higher donor capability show higher activity and productivity. Additionally, 11e was applied in the coupling of 4-chloro toluene with phenylboronic acid showing an excellent catalytic performance when compared to catalysts used by Fu, Beller and Buchwald. Furthermore, 11e is eligible for the synthesis of sterically hindered biaryls under mild reaction conditions. C–C Coupling reactions with the phosphino imidazolium salts 17b and 20 in ionic liquids [BMIM][PF6] and [BDMIM][BF4] were performed, showing less activity than in common organic solvents.
Se)R′2-4,5-R2-cC3N2 (11a-Se–f-Se; X = Br, I; R = H, Me; R′ = C6H5, cC6H11, cC4H3O) are reported. The structures of 11a-Se and [(1-(4-Br-C6H4)-cC3H2N2-3-n-Bu)2PdI2] (19) in the solid state were determined. Cyclovoltammetric measurements were performed with the ferrocenyl-containing molecules 5 and 7 showing reversible redox events at E0 = 0.108 V (ΔEp = 0.114 V) (5) and E0 = 0.183 V (ΔEp = 0.102 V) (7) indicating that 7 is more difficult to oxidise. Imidazole oxidation does not occur up to 1.3 V in dichloromethane using [(n-Bu)4N][B(C6F5)4] as supporting electrolyte, whereas an irreversible reduction is observed between −1.2–−1.5 V. The phosphino imidazoles 11a–k and the imidazolium salts 17a,b and 20, respectively, were applied in the Suzuki C–C cross-coupling of 2-bromo toluene with phenylboronic acid applying [Pd(OAc)2] as palladium source. Depending on the electronic character of 11a–k, 17a,b and 20 the catalytic performance of the in situ generated catalytic active species can be predicted. As assumed, more electron-rich phosphines with their higher donor capability show higher activity and productivity. Additionally, 11e was applied in the coupling of 4-chloro toluene with phenylboronic acid showing an excellent catalytic performance when compared to catalysts used by Fu, Beller and Buchwald. Furthermore, 11e is eligible for the synthesis of sterically hindered biaryls under mild reaction conditions. C–C Coupling reactions with the phosphino imidazolium salts 17b and 20 in ionic liquids [BMIM][PF6] and [BDMIM][BF4] were performed, showing less activity than in common organic solvents.
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CFc, R = H; 9, X = C6H5, R = Me; Fc = Fe(η5-C5H4)(η5-C5H5)), phosphino imidazoles 1-(4-X-C6H4)-2-PR′2-4,5-R2-cC3N2 (11a–k; X = Br, I, Fc, FcC
CFc, R = H; 9, X = C6H5, R = Me; Fc = Fe(η5-C5H4)(η5-C5H5)), phosphino imidazoles 1-(4-X-C6H4)-2-PR′2-4,5-R2-cC3N2 (11a–k; X = Br, I, Fc, FcC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C, Ph; R = H, Me; R′ = Ph, cC6H11, cC4H3O), imidazolium salts [1-(4-X-C6H4)-3-R′′-4,5-R2-cC3HN2]I (16a; X = Br, R = H, R′′ = n-Bu; 16b, X = Br, R = H, R′′ = n-C8H17; 16c, X = I, R = Me, R′′ = n-C8H17, 16d, X = H, R = Me, R′′ = n-C8H17) and phosphino imidazolium salts [1-C6H5-2-PR′2-3-n-C8H17-4,5-Me2-cC3N2]PF6 (17a, R′ = C6H5; 17b, R′ = cC6H11) or [1-(4-P(C6H5)2-C6H4)-3-n-C8H17-4,5-Me2-cC3HN2]PF6, (20) and their selenium derivatives 1-(4-X-C6H4)-2-P(
C, Ph; R = H, Me; R′ = Ph, cC6H11, cC4H3O), imidazolium salts [1-(4-X-C6H4)-3-R′′-4,5-R2-cC3HN2]I (16a; X = Br, R = H, R′′ = n-Bu; 16b, X = Br, R = H, R′′ = n-C8H17; 16c, X = I, R = Me, R′′ = n-C8H17, 16d, X = H, R = Me, R′′ = n-C8H17) and phosphino imidazolium salts [1-C6H5-2-PR′2-3-n-C8H17-4,5-Me2-cC3N2]PF6 (17a, R′ = C6H5; 17b, R′ = cC6H11) or [1-(4-P(C6H5)2-C6H4)-3-n-C8H17-4,5-Me2-cC3HN2]PF6, (20) and their selenium derivatives 1-(4-X-C6H4)-2-P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se)R′2-4,5-R2-cC3N2 (11a-Se–f-Se; X = Br, I; R = H, Me; R′ = C6H5, cC6H11, cC4H3O) are reported. The structures of 11a-Se and [(1-(4-Br-C6H4)-cC3H2N2-3-n-Bu)2PdI2] (19) in the solid state were determined. Cyclovoltammetric measurements were performed with the ferrocenyl-containing molecules 5 and 7 showing reversible redox events at E0 = 0.108 V (ΔEp = 0.114 V) (5) and E0 = 0.183 V (ΔEp = 0.102 V) (7) indicating that 7 is more difficult to oxidise.
Se)R′2-4,5-R2-cC3N2 (11a-Se–f-Se; X = Br, I; R = H, Me; R′ = C6H5, cC6H11, cC4H3O) are reported. The structures of 11a-Se and [(1-(4-Br-C6H4)-cC3H2N2-3-n-Bu)2PdI2] (19) in the solid state were determined. Cyclovoltammetric measurements were performed with the ferrocenyl-containing molecules 5 and 7 showing reversible redox events at E0 = 0.108 V (ΔEp = 0.114 V) (5) and E0 = 0.183 V (ΔEp = 0.102 V) (7) indicating that 7 is more difficult to oxidise. 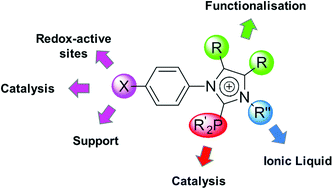

 Please wait while we load your content...
Please wait while we load your content...