Fixation of carbon dioxide by macrocyclic lanthanide(iii) complexes under neutral conditions producing self-assembled trimeric carbonato-bridged compounds with μ3-η2:η2:η2 bonding†‡
Abstract
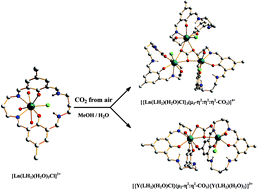
A series of mononuclear lanthanide(III) complexes [Ln(LH2)(H2O)3Cl](ClO4)2 (Ln = La, Nd, Sm, Eu, Gd, Tb, Lu) of the tetraiminodiphenolate macrocyclic ligand (LH2) in 95 : 5 (v/v) methanol–water solution fix atmospheric carbon dioxide to produce the carbonato-bridged trinuclear complexes [{Ln(LH2)(H2O)Cl}3(μ3-CO3)](ClO4)4·nH2O. Under similar conditions, the mononuclear YIII complex forms the dimeric compound [{Y(LH2)(H2O)Cl}(μ2-CO3){Y(LH2)(H2O)2}](ClO4)3·4H2O. These complexes have been characterized by their IR and NMR (1H, 13C) spectra. The X-ray crystal structures have been determined for the trinuclear carbonato-bridged compounds of NdIII, GdIII and TbIII and the dinuclear compound of YIII. In all cases, each of the metal centers are 8-coordinate involving two imine nitrogens and two phenolate oxygens of the macrocyclic ligand (LH2) whose two other imines are protonated and intramolecularly hydrogen-bonded with the phenolate oxygens. The oxygen atoms of the carbonate anion in the trinuclear complexes are bonded to the metal ions in tris-bidentate μ3-η2:η2:η2 fashion, while they are in bis-bidentate μ2-η2:η2 mode in the YIII complex. The magnetic properties of the GdIII complex have been studied over the temperature range 2 to 300 K and the magnetic susceptibility data indicate a very weak antiferromagnetic exchange interaction (J = −0.042 cm−1) between the GdIII centers (S = 7/2) in the metal triangle through the carbonate bridge. The luminescence spectral behaviors of the complexes of SmIII, EuIII, and TbIII have been studied. The ligand LH2 acts as a sensitizer for the metal ions in an acetonitrile–toluene glassy matrix (at 77 K) and luminescence intensities of the complexes decrease in the order Eu3+ > Sm3+ > Tb3+.

 Please wait while we load your content...
Please wait while we load your content...