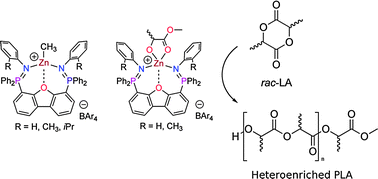
Cationic zinc complexes of a neutral pincer framework 4,6-(ArN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) PPh2)-dibenzofuran (L111: Ar = 2-iPrPh; L22: Ar = o-tolyl; L33: Ar = Ph), have been prepared and characterized. Crystallographic and NMR studies of the methylzinc complexes [LZnCH3+][BAr4−] (4a–6a: Ar = m-(CF3)2–C6H3; 4b–6b: Ar = Ph) demonstrated that the steric demands of the ligand dramatically affect the solid-state geometry. The cationic zinc–lactate complexes [LZnOR+][B(m-(CF3)2–C6H3)4−] (7: L = L22; 8: L = L33; R = CH(Me)CO2Me) were also studied, and their efficacy as lactide polymerization catalysts was examined. Polymerization using 7 requires heating to 60 °C, while complex 8 displays high activity at ambient temperature. The difference in activity can be attributed to κ2versus κ3 binding modes of the ligand, providing important insight into structure–activity relationships for this system. Complex 8 gives modestly heteroenriched PLA (Pr = 0.70), which represents the best stereocontrol yet achieved by a cationic metal catalyst.
PPh2)-dibenzofuran (L111: Ar = 2-iPrPh; L22: Ar = o-tolyl; L33: Ar = Ph), have been prepared and characterized. Crystallographic and NMR studies of the methylzinc complexes [LZnCH3+][BAr4−] (4a–6a: Ar = m-(CF3)2–C6H3; 4b–6b: Ar = Ph) demonstrated that the steric demands of the ligand dramatically affect the solid-state geometry. The cationic zinc–lactate complexes [LZnOR+][B(m-(CF3)2–C6H3)4−] (7: L = L22; 8: L = L33; R = CH(Me)CO2Me) were also studied, and their efficacy as lactide polymerization catalysts was examined. Polymerization using 7 requires heating to 60 °C, while complex 8 displays high activity at ambient temperature. The difference in activity can be attributed to κ2versus κ3 binding modes of the ligand, providing important insight into structure–activity relationships for this system. Complex 8 gives modestly heteroenriched PLA (Pr = 0.70), which represents the best stereocontrol yet achieved by a cationic metal catalyst.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) PPh2)-dibenzofuran (L111: Ar = 2-iPrPh; L22: Ar =
PPh2)-dibenzofuran (L111: Ar = 2-iPrPh; L22: Ar = 

 Please wait while we load your content...
Please wait while we load your content...