Tautomerization in the formation and collision-induced dissociation of alkali metal cation-cytosine complexes†
Abstract
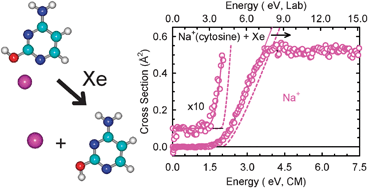
Noncovalent interactions between alkali metal cations and the various low-energy tautomeric forms of cytosine are investigated both experimentally and theoretically. Threshold collision-induced dissociation (CID) of M+(cytosine) complexes with Xe is studied using guided ion beam tandem mass spectrometry, where M+ = Li+, Na+, and K+. In all cases, the only dissociation pathway observed corresponds to endothermic loss of the intact cytosine molecule. The cross-section thresholds are interpreted to yield 0 and 298 K bond dissociation energies (BDEs) for the M+(cytosine) complexes after accounting for the effects of multiple ion-neutral collisions, the kinetic and internal energy distributions of the reactants, and dissociation lifetimes. Ab initio calculations are performed at the MP2(full)/6-31G* level of theory to determine the structures of the neutral cytosine tautomers, the M+(cytosine) complexes, and the TSs for unimolecular tautomerization. The molecular parameters derived from these structures are employed for the calculation of the unimolecular rates for tautomerization and the thermochemical analysis of the experimental data. Theoretical BDEs of the various M+(cytosine) complexes and the energy barriers for the unimolecular tautomerization of these complexes are determined at MP2(full)/6-311+G(2d,2p) level of theory using the MP2(full)/6-31G* optimized geometries. In addition, BDEs for the Li+(cytosine) complexes are also determined at the G3 level of theory. Based upon the tautomeric mixture generated upon thermal vaporization of cytosine, calculated M+–cytosine BDEs and barriers to tautomerization for the low-energy tautomeric forms of M+(cytosine), and measured thresholds for CID of M+(cytosine) complexes, we conclude that tautomerization occurs during both complex formation and CID.

 Please wait while we load your content...
Please wait while we load your content...