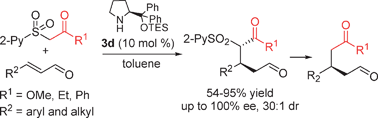
Synthesis of 3-substituted 1,5-aldehyde estersvia an organocatalytic highly enantioselective conjugate addition of new carbonylmethyl 2-pyridinylsulfone to enals†‡
Abstract
A highly enantioselective organocatalytic protocol for
- This article is part of the themed collection: Organocatalysis

 Please wait while we load your content...
Please wait while we load your content...