1,1-Carboboration
Abstract
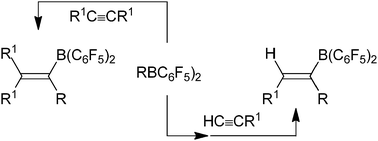
The use of very electrophilic ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CR undergo this reaction with the RB(C6F5)2 reagents rapidly under mild conditions to give high yields of very useful new alkenylborane products. Even internal
CR undergo this reaction with the RB(C6F5)2 reagents rapidly under mild conditions to give high yields of very useful new alkenylborane products. Even internal ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CR undergo 1,1-carboboration with the RB(C6F5)2 reagents to provide a novel way of carbon–carbon σ-bond
CR undergo 1,1-carboboration with the RB(C6F5)2 reagents to provide a novel way of carbon–carbon σ-bond

 Please wait while we load your content...
Please wait while we load your content...