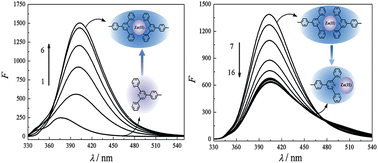
Pyrophosphate ion (PPi) is crucial in varieties of biological processes and industrial applications, and thus it is very important how to recognize it with high selectivity. In this contribution, one terpyridine (tpy)-based fluorescent molecule, 4-(methylphenyl)-2,2′:6′,2′′-terpyridine (mptpy), has been reported to display a highly selective recognition for PPi in the presence of Zn(II). After exposure toward the Zn(II) ion, the characteristic emission of mptpy at 376 nm red-shifted to 406 nm with a strong enhancement upon an excitation at 280 nm, and then blue-shifted to 388 nm with the further addition of PPi. Absorption and fluorescence measurements showed that other phosphates including phosphate (Pi) as well as nucleotide triphosphates could not induce the spectral changes similar to PPi, demonstrating the unique binding effect between mptpy-Zn(II) and PPi. This process could also discriminate PPi from other inorganic anions. Therefore, a tpy-based fluorescence method for the highly selective recognition of PPi could be developed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...