Abstract
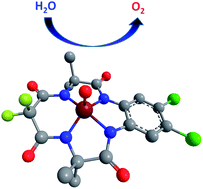
Theoretical models are used to demonstrate a catalytic cycle for the generation of O2 from an iron(III)-centered tetraamido macrocycle in water that is consistent with experimentally observed energetics in the presence of a sacrificial oxidant. Application of density functional theory and multireference second-order perturbation theory indicates that two proton-coupled electron transfer steps followed by an electron-transfer step first generate a reactive species that is well described as an iron(V)-oxo supported by a macrocycle that has also suffered a one-electron oxidation. Subsequent O–O bond formation occurs through water nucleophilic attack on the iron-oxo with the local solvent shell serving to relay a proton away from the reacting partners. Subsequent steps of proton-coupled electron transfer and low-energy water displacement of O2 complete the catalytic cycle. Modification of the TAML ligand to reduce the likely instability of the non-innocent aromatic radical may prove useful in future catalyst design.

 Please wait while we load your content...
Please wait while we load your content...