Organocatalytic Mannich/cyclization/aromatization sequence: direct synthesis of substituted pyrrole-3-carboxaldehydes†‡
Abstract
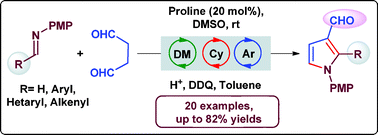
A robust method for the synthesis of substituted pyrrole-3-carboxaldehydes from N-PMP aldimines and succinaldehyde is reported. This reaction involves an organocatalytic direct Mannich reaction, and an acid catalyzed cyclization and oxidative aromatization sequence with high yields (up to 82%).

 Please wait while we load your content...
Please wait while we load your content...