Metal-free click polymerization of propiolates and azides: facile synthesis of functional poly(aroxycarbonyltriazole)s†
Abstract
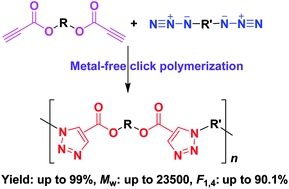
Azide–alkyne click polymerization is generally catalyzed by the Cu(I) complex, in which the removal of metallic residues after reaction remains challengeable. In this work, a metal-free click polymerization method was developed to avoid this problem. The diazide monomers (9 and 10) could readily react with aromatic and aliphatic dipropiolates (1 and 2) under simple heating to produce functional poly(aroxycarbonyltriazole)s (PACTs), a kind of polyester, with high molecular weights (Mw up to 23 500) and regioregularities (fraction of 1,4-disubstituted 1,2,3-triazole in polymers up to ∼90%) in excellent yields (up to ∼99%). The metal-free click polymerization also propagates smoothly in an open atmosphere without protection from oxygen and moisture. The PACTs are thermally stable, with 5% loss of their weights at temperatures higher than 335 °C. Thanks to the contained ester groups, the polymers can be rapidly degraded in the presence of potassium hydroxide aqueous solution. The PACT containing tetraphenylethene units exhibits a unique feature of aggregation-induced emission and can function as a fluorescent chemosensor to detect explosives in a superamplified manner.

 Please wait while we load your content...
Please wait while we load your content...