Abstract
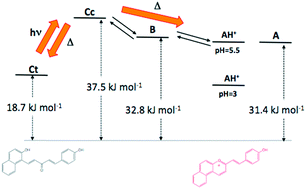
A new photochromic system based on 2-(4-hydroxystyryl)-1-naphthopyrylium encompasses the properties of the previously described naphthoflavylium and styrylflavylium systems. The photoproduct exhibits a colour deep in hue and is red shifted in comparison with the equivalent flavylium system. Reaction of 2-hydroxy-1-naphthaldehyde with p-hydroxystyrylmethylketone in acetic acid in the presence of tetrafluoroboric acid and acetic anhydride as catalysts leads to a mixture of two compounds: the photochromic 2-(4-hydroxystyryl)-1-naphthopyrylium and a second product 2-(4-acetoxystyryl)-1-naphthopyrylium resulting from the acetylation by acetic anhydride of the former. In acidic medium and at room temperature the hydrolysis of the acetoxy derivative leads to the 2-(4-hydroxystyryl)-1-naphthopyrylium, in circa 2 days, [HCl] = 0.25 M. The pH dependent chemical reactions taking place with 2-(4-hydroxystyryl)-1-naphthopyrylium were determined by UV-Vis, stooped flow, flash photolysis and 1H NMR and follow the same general pattern of flavylium derivatives. In order to rationalize the photochromism, an energy level diagram summarizing all the equilibrium and rate constants of the network was drawn.
- This article is part of the themed collection: In honour of Jean-Pierre Desvergne

 Please wait while we load your content...
Please wait while we load your content...