Pyridostatin analogues promote telomere dysfunction and long-term growth inhibition in human cancer cells†
Abstract
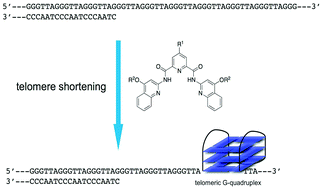
The synthesis, biophysical and biological evaluation of a series of G-quadruplex interacting small molecules based on a N,N′-bis(quinolinyl)pyridine-2,6-dicarboxamide scaffold is described. The synthetic analogues were evaluated for their ability to stabilize telomeric G-quadruplex DNA, some of which showed very high stabilization potential associated with high selectivity over double-stranded DNA. The compounds exhibited growth arrest of cancer cells with detectable selectivity over normal cells. Long-time growth arrest was accompanied by senescence, where telomeric dysfunction is a predominant mechanism together with the accumulation of restricted DNA damage sites in the genome. Our data emphasize the potential of a senescence-mediated anticancer therapy through the use of G-quadruplex targeting small molecules based on the molecular framework of pyridostatin.
- This article is part of the themed collection: Nucleic acids: new life, new materials

 Please wait while we load your content...
Please wait while we load your content...