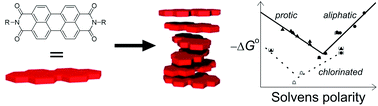
A series of six perylene bisimides (PBIs) with hydrophilic and hydrophobic side chains at the imide nitrogens were applied for a comparative study of the solvent and structural effects on the aggregation behaviour of this class of dyes. A comparison of the binding constants in tetrachloromethane at room temperature revealed the highest binding constant of about 105 M−1 for a PBI bearing 3,4,5-tridodecyloxyphenyl substituents at the imide nitrogens, followed by 3,4,5-tridodecylphenyl and alkyl-substituted PBIs, whereas no aggregation could be observed in the accessible concentration range for PBIs equipped with bulky 2,6-diisopropylphenyl substituents at the imide nitrogens. The aggregation behaviour of three properly soluble compounds was investigated in 17 different solvents covering a broad polarity range from nonpolar n-hexane to highly polar DMSO and water. Linear free energy relationships (LFER) revealed a biphasic behaviour between Gibbs free energies of aggregation and common empirical solvent polarity scales indicating particularly strong π–π stacking interactions in nonpolar aliphatic and polar alcoholic solvents whilst the weakest binding is observed in dichloromethane and chloroform. Accordingly, PBI aggregation is dominated by electrostatic interactions in nonpolar solvents and by solvophobic interactions in protic solvents. In water, the aggregation constant is increased far beyond LFER expectations pointing at a pronounced hydrophobic effect.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...