Bioinspired organocatalytic asymmetric reactions
Abstract
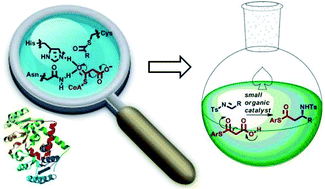
Several small organic molecule catalysts are reminiscent of natural enzymes in their mode of action and substrate interaction/activation. This striking similarity has been a great source of inspiration for the development of new organocatalytic asymmetric processes. A few representative examples, mostly dealing with catalysts interacting through multiple hydrogen-bonds (synthetic oxyanion holes), are highlighted in this perspective.

 Please wait while we load your content...
Please wait while we load your content...