Detrimental cationic doping of titania in photocatalysis: why chromium Cr3+-doping is a catastrophe for photocatalysis, both under UV- and visible irradiations
Abstract
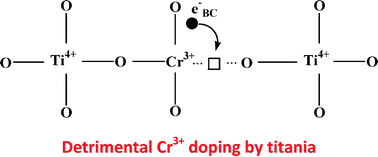
The chromium-doping of titania, initiated by electrochemists, was subsequently applied to photocatalysis to tentatively photo-sensitize titania in the visible region. The results were not conclusive and disparate. Presently, the photo-electronic behavior of pristine TiO2 and of Cr-doped (0.85 at%) TiO2 has been followed by UV-spectroscopy combined with electronic photoconductivity under vacuum and under oxygen pressure. Although Cr-doped (0.85 at%) TiO2 absorbs in the visible region, it only becomes a photoconductor through band-gap illumination and the doping causes a considerable decrease in photoconductivity by three orders of magnitude. Correspondingly, its photocatalytic activity was a disaster, confirmed by five different reactions representative of various media in which titania is active: (i) gas phase oxidations of

 Please wait while we load your content...
Please wait while we load your content...