Probing of bismuth antiulcer drug targets in H. pylori by laser ablation-inductively coupled plasma mass spectrometry
Abstract
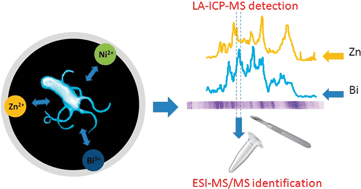
A method that allows partial denaturation of
* Corresponding authors
a Department of Chemistry, The University of Hong Kong, Hong Kong, P. R. China
b
LCABIE, UMR 5254, Hélioparc, 2, Av. Président Angot, 64053 Pau, France
E-mail:
joanna.szpunar@univ-pau.fr
c Warsaw University of Technology, ul. Naukowskiego 3, 00-664 Warsaw, Poland
A method that allows partial denaturation of

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
