Synthesis and evaluation of N-(2,3-dihydroxypropyl)-PEIs as efficient vectors for nucleic acids†
Abstract
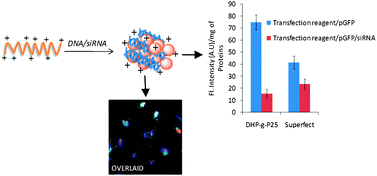
Branched polyethylenimine (bPEI, 25 kDa) has been widely used as an efficient delivery vector for nucleic acids in vitro. However, its charge-associated toxicity has limited its in vivo applications. In an attempt to control its toxicity, it was reacted with varying amounts of glycidol (2,3-epoxy-1-propanol) to obtain a small series of hydrophilic polymers, 2,3-dihydroxypropyl-grafted-polyethylenimines (DHP-g-P). The resulting polymers were characterized by 1H-NMR and subjected to interaction with negatively charged pDNA, which yielded complexes in the size range of ∼171–190 nm with a zeta potential of ∼+33–39 mV. Acid–base titration revealed no effect of substitution on the buffering capacity of the modified polymers. Grafting of 2,3-dihydroxypropyl groups on bPEI significantly improved the cell viability (i.e. almost non-toxic) as well as the DNA release properties of these modified polymers compared to native bPEI. Formation of a relatively loose DHP-g-P25/pDNA complex (the best working system in terms of transfection efficiency) resulted in the efficient nuclear release of pDNA for transcription, a prerequisite for efficient transfection. Subsequently, upon evaluation of their ability to transfer nucleic acids in vitro, the DHP-g-P/pDNA complexes exhibited higher gene transfection efficiency with one of the formulations, DHP-g-P25/DNA complex, displaying ∼2.7 folds higher GFP expression than bPEI and ∼2.3–3.5 folds higher than the selected commercial transfection reagents used in this study. Further to quantify the extent of GFP positive cells, FACS analysis was performed, which revealed DHP-g-P25/DNA mediated gene expression in ∼51% cells outcompeting bPEI, Superfect™, Fugene™ and Lipofectamine™. Sequential delivery of GFP-specific siRNA resulted in ∼78% suppression of the target gene compared to ∼49% achieved by Fugene™. All these results demonstrate the potential of these polymers for in vivo gene delivery.

 Please wait while we load your content...
Please wait while we load your content...