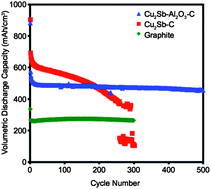
Cu2Sb–Al2O3–C nanocomposite alloy material has been synthesized via a mechanochemical reduction of Sb2O3 with Al and Cu metals in the presence of carbon (acetylene black). X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM), and scanning electron microscopy (SEM) reveal that these nanocomposites are composed of nanostructured Cu2Sb particles in an amorphous Al2O3 and conductive carbon matrix. The presence of Al2O3 in the nanocomposite drastically improves the cycle life of the Cu2Sb–C material and enables a remarkable capacity retention of 80% over 500 cycles (330 mAh/g) after the first cycle. With a tap density of ∼1.4 g cm−3, Cu2Sb–Al2O3–C offers significantly higher volumetric capacity than the commonly used graphite anode. A conductive Cu framework supports the electrochemically-active Sb and results in low impedance and good rate capability, while the matrix of ceramic oxide and conductive carbon reduce the agglomeration of the nanocrystalline Cu2Sb particles and buffer the volume expansion/contraction that occurs during cycling. TEM of the electrodes that were cycled between 1 and 500 cycles shows the evolution of the morphology of Cu2Sb particles in the presence and absence of Al2O3. Because the Cu2Sb–Al2O3–C nanocomposite electrodes can operate well above the potential where lithium plating occurs, it is viewed as a safer alternative to graphite anodes.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...