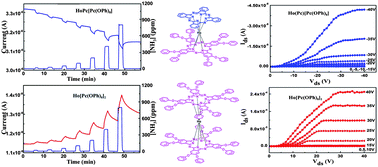
The semiconducting properties of the heteroleptic and homoleptic bis(phthalocyaninato) holmium complexes bearing electron-withdrawing phenoxy substituents at the phthalocyanine periphery, namely Ho(Pc)[Pc(OPh)8] (1) and Ho[Pc(OPh)8]2 (2) [Pc = unsubstituted phthalocyaninate; Pc(OPh)8 = 2,3,9,10,16,17,23,24-octaphenoxyphthalocyaninate] have been investigated comparatively. Using a solution-based Quasi–Langmuir–Shäfer (QLS) method, the thin solid films of the two compounds were fabricated. The structure and properties of the thin films were investigated by UV-vis absorption spectra, X-ray diffraction (XRD) and atomic force microscopy (AFM). Experimental results indicated that H-type molecular stacking mode with the common preferential molecular “edge-on” orientation relative to the substrate has been formed, and the intermolecular face-to-face π–π interaction and film microstructures are effectively improve by increasing the number of phenoxy substituents of the Pc periphery within the double-decker complexes. The electrical conductivity of Ho(Pc)[Pc(OPh)8] films was measured to be approximately 4 orders of magnitude larger than that of Ho[Pc(OPh)8]2 films, indicating significant effect of peripheral electron-withdrawing phenoxy groups on conducting behaviour of bis(phthalocyaninato) holmium complexes. In addition, the gas sensing behaviour of the QLS films of 1 and 2 toward electron donating gas, NH3, was investigated in the concentration range of 15–800 ppm. Surprisingly, contrary responses towards NH3 were found for the QLS films of 1 and 2. In the presence of NH3, the conductivity of the films of Ho(Pc)[Pc(OPh)8] (1) decreased while the conductivity of the films of Ho[Pc(OPh)8]2 (2) increased. This observation clearly demonstrated the p- and n-type semiconducting nature for 1 and 2, respectively. Furthermore, compared to the heteroleptic 1 having a hole mobility of 1.7 × 10−4 cm2 V−1 s−1, homoleptic 2 exhibits an electron mobility as high as 0.54 cm2 V−1 s−1. Therefore, the inversion of the semiconducting nature of the double-deckers from p- to n-type can be successfully and easily realized just by increasing the number of peripheral phenoxy groups attached to the conjugated Pc cores.

 Please wait while we load your content...
Please wait while we load your content...